If you’ve been reading my Gene-of-the-Week posts for a while, you may recall that, although I know admittedly little about the immune system, I have a particular, personal interest in autoimmune diseases. Recently, my immune system has been aberrantly attacking my own healthy tissues, resulting in some really annoying joint pain and swelling. As much as it sucks, the more I learn about the immune system, the more I don’t blame it. I mean, there is so much going on in my body, how is my immune system really supposed to keep track of what is me and what isn’t?!
One way the immune system learns to differentiate self from non-self is through the help of our Gene-of-the-Week: the autoimmune regulator gene, or AIRE.
First: T cell development
T cells are produced in bone marrow and migrate to the thymus (hence the “T”), where they mature. In case you’ve forgotten about it, the thymus is the small organ hiding behind your sternum. The thymus is divided into a cortex (outer layer) and a medulla (middle). Culinary thymus is euphemistically known as “sweetbread.”
Overshadowed by such organs as the heart and lungs, the thymus is seriously underappreciated!! (Image from Wikipedia)
The maturation of T cells is marked by their expression of CD4 and CD8 as they move from the cortex into the medulla and out of the thymus. Immature T cells (thymocytes) from bone marrow are double-negative for CD4 and CD8 (CD4–/CD8–). In the cortex of the thymus, thymocytes become double-positive (CD4+/CD8+). In a single day, the mouse thymus can generate up to 50 million double-positive thymocytes. Wow! BUT, it’s hard out there for a thymocyte. 90% of these double-positive cells undergo “death by neglect” in the cortex.1
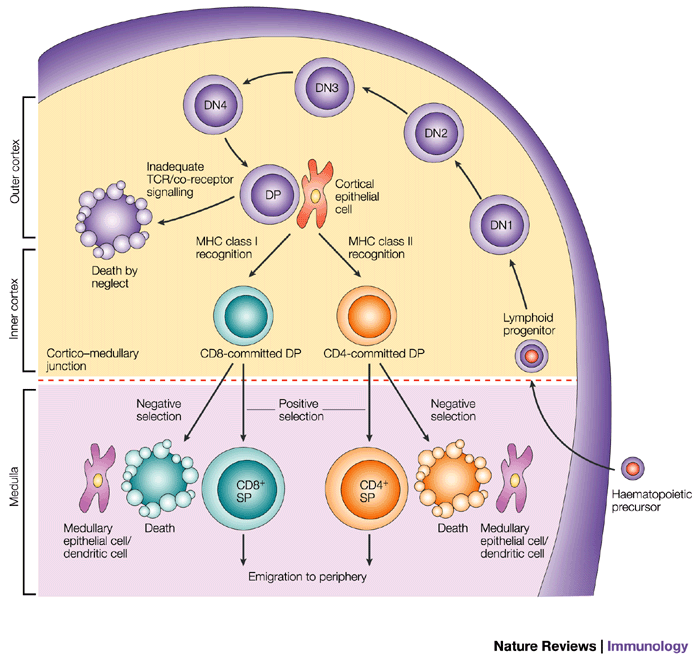
A schematic of T cell development in the thymus, from Germain, 2002.2
In the cortex, T cell receptors (TCRs) are generated randomly via V(D)J recombination. TCRs sit on the outside of the thymocytes and enable interaction with cortical thymic epithelial cells (cTECs), which are antigen-presenting cells (APCs). Your cTECs present self-antigens (i.e., antigens originating from your body) on major histocompatibility complexes (MHCs). If the affinity of a thymocyte’s TCR to the presented antigen is too weak, the thymocyte will undergo death by neglect. If affinity of the TCR to the self-antigen is too great, negative selection leads to apoptosis. Therefore, only cells with intermediate affinities for self-antigens are “positively selected” to mature into single-positive (CD4+/CD8– or CD4–/CD8+) cells. Cells that interact with MHC class II will become CD4+, and cells that interact with MHC class I will become CD8+.
Interestingly, processing in the cortex does not select against thymocytes that will cause autoimmunity. That’s is where AIRE comes into play.
The importance of AIRE
If T cells mature in the thymus, then how do they learn not to react to tissue-specific antigens? Rather than migrating from other organs into the thymus, the tissue-restricted antigens (TRAs) are “promiscuously” expressed in medullary thymic epithelial cells (mTECs). At any given time, all TRAs are expressed in the medulla of the thymus. Expression of TRAs is controlled, of course, by AIRE.

Nelly Furtado is obviously the personification of a thymically-expressed TRA. And, to unnecessarily extend the metaphor, the promiscuous boy is a thymocyte interacting with all those other mTECs… smh.
AIRE binds to non-methylated histone 3 lysine 4 (H3K4), which marks promoters in closed chromatin regions—closed, because they aren’t supposed to be expressed!3 Although each TRA is only expressed in 1-3% of mTECs, the single-positive thymocytes that make it through the cortex will hang out in the medulla for about 5 days.4 During this time, they move about and encounter several hundreds of mTECs expressing various TRAs. In addition, it was recently found that mTECs share their TRAs with other mTECs, as well as with dendritic cells (DCs).5 The combinations of shared TRAs and cell migration means that all thymocytes will eventually interact with every TRA! Thymocytes that react strongly to TRAs are negatively selected to undergo apoptosis, or they are redirected to become regulatory T (TReg) cells, which maintain tolerance. In addition to TReg cells, the remaining survivors of positive and negative selection leave the thymus as naïve T cells.
Lack of AIRE
Studies using mice have demonstrated that increased Aire expression up-regulates TRA expression, while Aire-deficiency prevents TRA expression. 6 Humans with AIRE defects may present with a rare recessive disorder called autoimmune polyendocrinopathy-candidiasis-ectodermal dystrophy (APECED), aka autoimmune polyendocrine syndrome type 1 (APS-1). Clinical features of APECED/APS-1 include hypoparathyroidism (hypocalcaemia), yeast infection, and adrenal insufficiency (Addison’s disease).7 Of particular interest to the author, polymorphisms in AIRE may also lead to rheumatoid arthritis in Japanese and Chinese populations.89
Maybe I just need some AIRE!
(…But not too much: a recent study found that increased expression of AIRE in patients with down syndrome (DS) may contribute to a relatively frequent autoimmune DS phenotype!10).
References:
AIRE has always been popular, but a recent finding by Keane et al. (2014) that AIRE may also increase tissue-specific splicing of TRAs in the thymus may have further increased its popularity.11
- Klein, L., Kyewski, B., Allen, P.M., and Hogquist, K. a (2014). Positive and negative selection of the T cell repertoire: what thymocytes see (and don’t see). Nat. Rev. Immunol. 14, 377–391. [↩]
- Germain, R.N. (2002). T-cell development and the CD4-CD8 lineage decision. Nat. Rev. Immunol. 2, 309–322. [↩]
- Zumer, K., Saksela, K., and Peterlin, B.M. (2013). The mechanism of tissue-restricted antigen gene expression by AIRE. J. Immunol. 190, 2479–2482. [↩]
- Klein, L., Kyewski, B., Allen, P.M., and Hogquist, K. a (2014). Positive and negative selection of the T cell repertoire: what thymocytes see (and don’t see). Nat. Rev. Immunol. 14, 377–391. [↩]
- Perry, J.S.A., Lio, C.-W.J., Kau, A.L., Nutsch, K., Yang, Z., Gordon, J.I., Murphy, K.M., and Hsieh, C.-S. (2014). Distinct Contributions of Aire and Antigen-Presenting-Cell Subsets to the Generation of Self-Tolerance in the Thymus. Immunity 41, 414–426. [↩]
- Eldershaw, S.A., Sansom, D.M., and Narendran, P. (2011). Expression and function of the autoimmune regulator (Aire) gene in non-thymic tissue. Clin. Exp. Immunol. 163, 296–308. [↩]
- Kisand, K., Peterson, P., and Laan, M. (2014). Lymphopenia-induced proliferation in aire-deficient mice helps to explain their autoimmunity and differences from human patients. Front. Immunol. 5, 51. [↩]
- Terao, C., Yamada, R., Ohmura, K., Takahashi, M., Kawaguchi, T., Kochi, Y., Okada, Y., Nakamura, Y., Yamamoto, K., Melchers, I., et al. (2011). The human AIRE gene at chromosome 21q22 is a genetic determinant for the predisposition to rheumatoid arthritis in Japanese population. Hum. Mol. Genet. 20, 2680–2685. [↩]
- Shao, S., Li, X.-R., Cen, H., and Yin, Z.-S. (2014). Association of AIRE polymorphisms with genetic susceptibility to rheumatoid arthritis in a Chinese population. Inflammation 37, 495–499. [↩]
- Giménez-Barcons, M., Casteràs, A., Armengol, M.D.P., Porta, E., Correa, P. a, Marín, A., Pujol-Borrell, R., and Colobran, R. (2014). Autoimmune predisposition in Down syndrome may result from a partial central tolerance failure due to insufficient intrathymic expression of AIRE and peripheral antigens. J. Immunol. 193, 3872–3879. [↩]
- Keane, P., Ceredig, R., and Seoighe, C. (2014). Promiscuous mRNA splicing under the control of AIRE in medullary thymic epithelial cells. Bioinformatics 1–5. [↩]
