Some Like It Hot — #GeneOTW with TRPV1
The transient receptor potential cation channel subfamily V member 1, or TRPV1, is most commonly known as the capsaicin receptor.1 Capsaicin is the compound that makes chili peppers “hot,” and the TRPV1 receptor encoded by TRPV1 is what makes us sense the heat. When capsaicin binds to the receptor, cations (most notably Ca2+) can flow into the cell, and then… HOT!

A Trpv1 knockout mouse eats a habanero chili! It’s all like, “No big deal.” (Image from The Journal of Neuroscience)
But sensing spice isn’t the only role of TRPV1 receptors. They also sense noxious heat (>109°F, ouch!), low (acidic) pH, and allyl isothiocyanate from wasabi, mustard, and horseradish. The receptor is also important for regulating body temperature and plays a key role in the pain pathway.2 The role of the TRPV1 receptor in pain makes it a major target for the development of analgesics, but its role in body temperature regulation means that the use of TRPV1 antagonists can cause hyperthermia, so you’ll be in less pain but also hot… and not in a good way!3 Interestingly, TRPV1 has also been implicated in depression, schizophrenia, and other psychiatric disorders (which are my jam) through the dopaminergic and cannabinoid systems.4
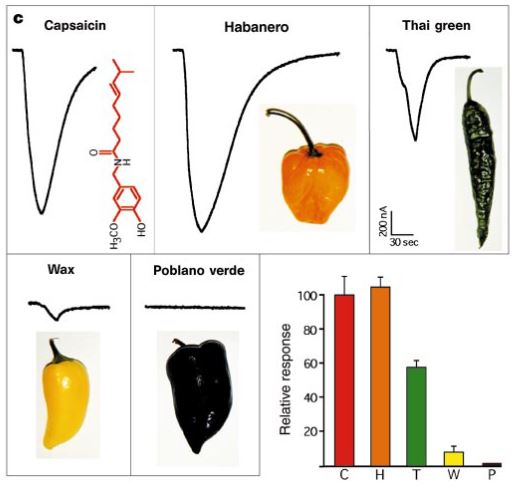
Responses to capsaicin and extracts from four varieties of peppers in oocytes expressing TRPV1 (30 s application). From Caterina et al. (1997).
TRPV1 receptors are also activated by alcohol. When Trpv1 is knocked out in mice, the mice have a greater preference for and consume more ethanol than wildtype mice. They are also less sensitive to ethanol-induced sedation and recover faster from ethanol-induced motor incoordination (which is good if they’re drinking so much!).5 Recently, a study shared by our faithful follower @usethespacebar reported that single nucleotide polymorphisms (SNPs) in TRPV1 mediate the perceived bitterness and burning sensation of ethanol.6 Unfortunately, the authors did not report the subjects’ enjoyment and consumption of alcoholic beverages outside the lab, but presumably these SNPs would effect both (though variation enjoyment of the bitterness and burning would probably complicate the analysis…).
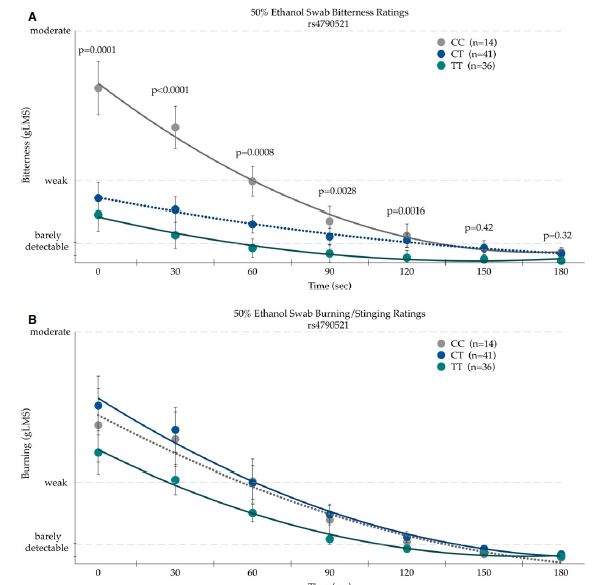
The TRPV1 SNP rs4790521 influences the bitterness and burning sensation experienced when ethanol is swabbed onto the circumvallate papillae (at the back of the tongue) for 30 seconds. From Allen et al. (2008).
Clearly, TRPV1 is doing a whole heck of a lot in our bodies. I had no idea it was doing so much! This brief post gives only a small glimpse into the world of TRPV1 science. I hope it’s just enough to whet your appetite and you’ll fall into the rabbit hole of TRPs and other ion channels. And maybe you can ask @channelomics for more info 😉
Do you have a suggestion for a Gene-of-the-Week? If so, let us know!
References
- Caterina, M.J., Schumacher, M.A., Tominaga, M., Rosen, T.A., Levine, J.D., and Julius, D. (1997). The capsaicin receptor: a heat-activated ion channel in the pain pathway. Nature 389, 816–824. [↩]
- Gavva, N.R., Bannon, A.W., Surapaneni, S., Hovland, D.N., Lehto, S.G., Gore, A., Juan, T., Deng, H., Han, B., Klionsky, L., et al. (2007). The vanilloid receptor TRPV1 is tonically activated in vivo and involved in body temperature regulation. J. Neurosci. 27, 3366–3374. [↩]
- Khairatkar-Joshi, N., and Szallasi, A. (2009). TRPV1 antagonists: the challenges for therapeutic targeting. Trends Mol. Med. 15, 14–22. [↩]
- Chahl, L.A. (2011). TRP Channels and Psychiatric Disorders. In Transient Receptor Potential Channels, M.S. Islam, ed. (Dordrecht: Springer Netherlands), pp. 987–1009. [↩]
- Blednov, Y.A., and Harris, R.A. (2009). Deletion of vanilloid receptor (TRPV1) in mice alters behavioral effects of ethanol. Neuropharmacology 56, 814–820. [↩]
- Allen, A.L., McGeary, J.E., and Hayes, J.E. (2014). Polymorphisms in TRPV1 and TAS2Rs Associate with Sensations from Sampled Ethanol. Alcohol. Clin. Exp. Res. 38, 2550–2560. [↩]
Recent Posts
What’s your type? — #GeneOTW with ABO
by Kerin Higa | Oct 13, 2014 | BioGPS, GeneOfTheWeek
I know what you’re thinking. This blog is degenerating. Last week, we talked about semen, and this week, we’re talking about finding the one. Right? “What’s your type?” sounds like the perfect BuzzFeed quiz for your...The Double Life of FAS – #GeneOTW
by Kerin Higa | Sep 29, 2014 | BioGPS, cancer, GeneOfTheWeek
1989 was an important year. In May of 1989, Yonehara et al. published a report that treatment with an anti-FAS antibody leads to the death of cells expressing our ominously-named gene-of-the-week, “Fas cell surface death receptor” (FAS).1 In July...This is my jam—#GeneOTW with ANK3
by Kerin Higa | Sep 15, 2014 | BioGPS, GeneOfTheWeek
Happy Monday, friends! Once a week since April, Melissa and I have blogged about popular genes that users have searched for using BioGPS. Based on user data, we have been led to explore a wide variety of genes in our Gene-of-the-Week...CHGB & AMFOG: Chromogranin B and adrenomedullary function of granins – #GeneOTW
by Kerin Higa | Sep 1, 2014 | BioGPS, GeneOfTheWeek
CBGB & OMFUG opened in 1973 as a venue for “Country, Bluegrass, and Blues & Other Music for Uplifting Gormandizers.” Despite its namesake aspirations, CBGB quickly became important for the creation, cultivation, and release of punk rock...