Sometimes, the entire month of December feels like an endless stream of holiday parties, non-stop feasting, and way too many cookies. Since my holiday vices tend toward roast duck and copious amounts of sticky rice, I’m definitely looking forward to a more fitness-focused January. With health-related New Year’s resolutions being so common, many people (myself included) will soon be trading in their food comas for sweat and sore muscles. But what actually happens to your muscles when you exercise?
As a mechanically active tissue, skeletal muscle is subject to a lot of stress—it’s been suggested that rigorous exercise can even cause tiny tears in your muscle fibers. Damage to the cell membrane can have disastrous results, what with important stuff leaking out, extra stuff sneaking in, and overall cell homeostasis being disrupted. Fortunately, holes in muscle cell membranes can be resealed in less than a minute!1 Generally, the membrane repair system initiates fusion of intracellular vesicles, which end up forming a patch over the lesion site.2 How this exactly happens still isn’t completely clear, but it is evident that our current Gene-of-the-Week, dysferlin, plays an important role.

Muscles are like a biological Russian nesting doll! Once you get down to a single muscle fiber cell, damage to the sarcolemma (or cell membrane) is repaired with help from dysferlin. [Image: OpenStax College]
Highly expressed in skeletal and cardiac muscle, dysferlin is a transmembrane protein that’s quickly recruited to damaged membrane. In fact, live cell imaging shows that dysferlin aggregates within seconds of a lesion, and forms a stable lattice-like structure over the wound.3 Once there, it’s thought to interact with other proteins implicated in membrane resealing. Dysferlin also binds phospholipids, including ones that are critical components of a cell membrane.4
Furthermore, dysferlin appears to be regulated by calcium; its phospholipid binding depends on calcium,5 as does its cleavage into the mini-dysferlin form that aggregates at membrane lesions.6 Especially since membrane damage results in an influx of extracellular calcium, it’s tempting to propose a scenario in which dysferlin acts as a calcium (and thus damage) sensor to initiate membrane resealing.7
Mutations in dysferlin can cause several types of muscular dystrophy, including Miyoshi myopathy and limb-girdle muscular dystrophy.8 All of these so-called dysferlinopathies (when dysferlin is absent or deficient) are recessively inherited, and result in progressive muscle weakness and atrophy. While muscle membrane repair is impaired in these patients, the muscle wasting is most likely caused by a pathogenic immune response.9 This suggests that dysferlin may have an additional (as yet unknown) role outside of membrane repair.
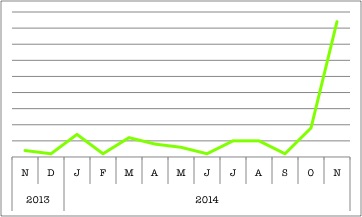
Dysferlin’s popularity on BioGPS is increasing, maybe because of a new paper that proposes a drug treatment for dysferlinopathies!
Even without all these details solved, there’s been some recent progress in finding disease treatments! Depending on the type of mutation, some patients may still have functional dysferlin—you just have to boost the amount that’s available. With missense mutations, a functional dysferlin might be made, but would be recognized as “wrong” by the cells’ quality control system. The missense mutated dysferlin would be subsequently degraded, even if it remained (mostly) functional. But, in a small trial with three patients, researchers were able to reduce dysferlin degradation with a proteasome inhibitor drug!10 While dysferlin levels did rise in these patients, long-term effect on disease symptoms and progression hasn’t been tested. Still, this is just the first step toward larger-scale clinical trials!
With so much unknown, dysferlin is still a rather mysterious protein. Regardless, the details might be hazy, but it’s clear that dysferlin is essential to muscle membrane repair… and other important, unknown functions. So this holiday season, when you’re crunching away on those cookies (and contemplating an exercise plan for 2015), just take a moment to reflect on dysferlin, and all the complicated repair machinery that keeps our muscles going. It’s certainly something worth toasting to!

Happy holidays from BioGPS (and three cheers for dysferlin)! [Image: Bill Masson]
REFERENCES:
- Allen DG, Whitehead NP, Yeung EW. (2005) Mechanisms of stretch-induced muscle damage in normal and dystrophic muscle: role of ionic changes. J Physiol 567(Pt 3):723-35. [↩]
- Mariano A, Henning A, Han R. (2013) Dysferlin-deficient muscular dystrophy and innate immune activation. FEBS J 280(17):4165-76. [↩]
- McDade JR, Archambeau A, Michele DE. (2014) Rapid actin-cytoskeleton-dependent recruitment of plasma membrane-derived dysferlin at wounds is critical formuscle membrane repair. FASEB J 28(8):3660-70. [↩]
- Mariano A, Henning A, Han R. (2013) Dysferlin-deficient muscular dystrophy and innate immune activation. FEBS J 280(17):4165-76. [↩]
- Mariano A, Henning A, Han R. (2013) Dysferlin-deficient muscular dystrophy and innate immune activation. FEBS J 280(17):4165-76. [↩]
- Redpath GM, Woolger N, Piper AK, Lemckert FA, Lek A, Greer PA, North KN, Cooper ST. (2014) Calpain cleavage within dysferlin exon 40a releases a synaptotagmin-like module for membrane repair. Mol Biol Cell 25(19):3037-48. [↩]
- Mariano A, Henning A, Han R. (2013) Dysferlin-deficient muscular dystrophy and innate immune activation. FEBS J 280(17):4165-76. [↩]
- Azakir BA, Erne B, Di Fulvio S, Stirnimann G, Sinnreich M. (2014) Proteasome inhibitors increase missense mutated dysferlin in patients with muscular dystrophy. Sci Transl Med 6(250):250ra112 [↩]
- Mariano A, Henning A, Han R. (2013) Dysferlin-deficient muscular dystrophy and innate immune activation. FEBS J 280(17):4165-76. [↩]
- Azakir BA, Erne B, Di Fulvio S, Stirnimann G, Sinnreich M. (2014) Proteasome inhibitors increase missense mutated dysferlin in patients with muscular dystrophy. Sci Transl Med 6(250):250ra112 [↩]
