1981 was an eventful year. The Iran Hostage Crisis had finally ended. Ronald Reagan became the 40th President of the United States, and shortly thereafter, survived an assassination attempt. In the soap opera General Hospital, the marriage of two beloved characters became (and remains today) the highest-rated episode in daytime television history. And on June 5th, the CDC reported 5 cases of a rare lung infection in young, previously healthy, gay men—all with apparent immune system deficiencies. This marked the beginning of the AIDS epidemic.

The NAMES Project AIDS Quilt was made in remembrance of loved ones that died from AIDS-related causes. It memorializes more than 94,000 people, and weighs an estimated 54 tons.
Thirty-three years later, HIV has infected at least 60 million people and caused over 25 million deaths.1 The latest estimate for number of people living with HIV is 35.3 million2—that’s more than the total population of Canada. This number is rising, but that’s a good sign because people with HIV are living longer. Global incidence (which reflects new cases of HIV infection) is decreasing—except in Europe and the Americas where the availability of anti-retroviral drugs has resulted in so-called “therapeutic optimism,” and thus a return to risky sexual behavior.3 In the last few decades, there’s been a very vocal, effective public campaign against HIV/AIDS. However, the history of war against HIV extends much further…

RNF115 is climbing the BioGPS popularity charts, with a newly published paper demonstrating (for the 1st time) that RNF115 is a restriction factor. In just a few weeks, this PloS article’s been viewed more than 1000 times!
The origin of modern-day HIV traces back nearly 100 years to South Cameroon, where a single transmission event of Simian Immunodeficiency Virus (SIV) crossed from chimpanzee to human, eventually mutating to become the primary strain of HIV driving today’s AIDS pandemic.4 The first humans to be infected were likely to be early 20th century bushmeat hunters, but primates have been battling SIV for tens of thousands of years. Locked in an evolutionary arms race with these retroviruses, host species have developed a complex system of “restriction factors” that suppress virus production at various stages of the virus life-cycle. Proteins that contribute towards this innate immunity to HIV are of intense interest for therapeutic strategy; one of the newest restriction factors to be discovered is encoded by the gene RNF115.
RNF115 encodes a RING finger protein (like #GeneOTW alumnus, PCGF2) that impairs the spread of viral particles through the host body.5 When HIV first targets to (and fuses with) immune cells, its genetic material is released into the host cell, and eventually incorporates with host cell DNA. In this manner, the host cell machinery can be hijacked into making HIV-specific proteins. These proteins get assembled into new HIV virions, which are then unleashed from the cell membrane, going on to infect other cells. RNF115 impairs the HIV replication process by preventing new virion assembly; once the new HIV-specific proteins are made, RNF115 binds and ubiquitinates them, marking them for degradation.6 Furthermore, RNF115 also impairs HIV virion release through a different pathway, in conjunction with tetherin (another restriction factor).7
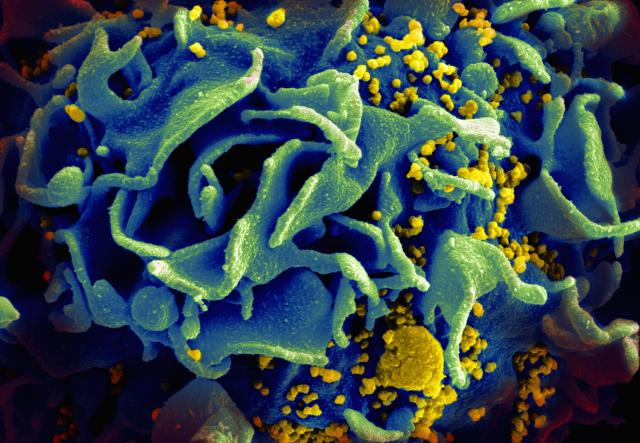
A scanning electron micrograph of an HIV-infected T cell. Newly replicated HIV virions (yellow) are budding off the cell, getting ready to infect other cells. RNF115 prevents the assembly and release of these viral particles. Credit: NIAID.
The first hope with discoveries such as these is that knowledge of genes like RNF115 can be translated into direct therapies against HIV. Of course it’s too early to speculate, but as with much of biology… it’s complicated. RNF115 goes by many other names, but the most commonly used one is BCA2, for Breast Cancer-Associated gene 2. In fact, this gene is actually up-regulated in more than 50% of breast cancers.8 So, increasing RNF115/BCA2 might not be the best strategy for developing an anti-retroviral drug. Even so, any insight into the mechanisms of HIV progression or innate immunity can still have far-reaching impact on future therapies—just look at how far we’ve come in the last thirty years.
References:
- Sharp PM, Hahn BH. (2011) Origins of HIV and the AIDS pandemic. Cold Spring Harb Perspect Med1(1):a006841. doi: 10.1101/cshperspect.a006841. [↩]
- Maartens G, Celum C, Lewin SR. (2014) HIV infection: epidemiology, pathogenesis, treatment, and prevention. Lancet pii: S0140-6736(14)60164-1. doi: 10.1016/S0140-6736(14)60164-1. [↩]
- Maartens G, Celum C, Lewin SR. (2014) HIV infection: epidemiology, pathogenesis, treatment, and prevention. Lancet pii: S0140-6736(14)60164-1. doi: 10.1016/S0140-6736(14)60164-1. [↩]
- Sharp PM, Hahn BH. (2011) Origins of HIV and the AIDS pandemic. Cold Spring Harb Perspect Med1(1):a006841. doi: 10.1101/cshperspect.a006841. [↩]
- Nityanandam R, Serra-Moreno R. (2014) BCA2/Rabring7 targets HIV-1 Gag for lysosomal degradation in a tetherin-independent manner. PLoS Pathog 10(5):e1004151. doi: 10.1371/journal.ppat.1004151. [↩]
- Nityanandam R, Serra-Moreno R. (2014) BCA2/Rabring7 targets HIV-1 Gag for lysosomal degradation in a tetherin-independent manner. PLoS Pathog 10(5):e1004151. doi: 10.1371/journal.ppat.1004151. [↩]
- Miyakawa K, Ryo A, Murakami T, Ohba K, Yamaoka S, Fukuda M, Guatelli J, Yamamoto N. (2009) BCA2/Rabring7 promotes tetherin-dependent HIV-1 restriction. PLoS Pathog5(12):e1000700. doi: 10.1371/journal.ppat.1000700. [↩]
- Nityanandam R, Serra-Moreno R. (2014) BCA2/Rabring7 targets HIV-1 Gag for lysosomal degradation in a tetherin-independent manner. PLoS Pathog 10(5):e1004151. doi: 10.1371/journal.ppat.1004151. [↩]
